 Emperra GmbH of Potsdam, Germany, is expanding its Esysta diabetes management product systems with what it says is the first Bluetooth insulin pen. The Esysta BT smart insulin pen logs injected insulin doses directly to the user’s digital blood glucose via the Bluetooth wireless interface, as well as being connected through the 868 MHz interface.
Emperra GmbH of Potsdam, Germany, is expanding its Esysta diabetes management product systems with what it says is the first Bluetooth insulin pen. The Esysta BT smart insulin pen logs injected insulin doses directly to the user’s digital blood glucose via the Bluetooth wireless interface, as well as being connected through the 868 MHz interface.
Both the system’s hardware components and software solutions (Esysta portal, Esysta app on Apple iOS and Android) have been certified as CE-compliant medical products, providing patients and doctors with a tool to enhance quality of medical care for type 1 and type 2 diabetics.
Currently approved for reimbursement by statutory health insurance companies of the German healthcare system, Emperra, a medical technology startup, has filed the smart insulin pen-based Esysta telemonitoring solution with the U.S. Food and Drug Administration (FDA) for U.S. market approval, and says FDA clearance is expected sometime this year.
 The Esysta BT pen allows for the direct transfer of insulin data to automatically keep a diary
The Esysta BT pen allows for the direct transfer of insulin data to automatically keep a diary
 “The smart insulin pen and corresponding software appeal to patients and doctors who prefer state-of-the-art technical solutions and who have recognized that the digitalization of diabetes management is a simple and reliable solution,” Emperra CEO Dr. Christian Krey said in a press release.
“The smart insulin pen and corresponding software appeal to patients and doctors who prefer state-of-the-art technical solutions and who have recognized that the digitalization of diabetes management is a simple and reliable solution,” Emperra CEO Dr. Christian Krey said in a press release.
Emperra also notes that by using sophisticated software solutions compliant with medical products and the highest integrated data security standards ISO/IEC 27001, several teams of doctors can for the first time use raw insulin data from smart pens to solve complex issues with patients whose insulin dosage levels are difficult to adjust telemedically.
 The Esysta iOS and Android apps play treatment recommendations back using innovative “traffic light algorithms.” Emperra reports that Esysta convinced most patients participating in a field trial conducted by German health insurance companies, as well as their doctors and caregivers, within the first few months of use.
The Esysta iOS and Android apps play treatment recommendations back using innovative “traffic light algorithms.” Emperra reports that Esysta convinced most patients participating in a field trial conducted by German health insurance companies, as well as their doctors and caregivers, within the first few months of use.
Emperra is currently the only company that can support all age groups of diabetic patients using smart insulin pens, with the largest cohort of older type 2 diabetics receiving insulin treatment being digitally connected to technology for the first time.
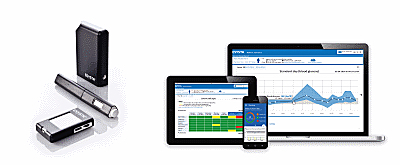
 ESYSTA: Web-based telemonitoring system consisting of a base unit, insulin pen, blood glucose meter, app, and online portal.
ESYSTA: Web-based telemonitoring system consisting of a base unit, insulin pen, blood glucose meter, app, and online portal.
 “We are proud that we can use all U-100 insulins in 3 ml cartridges with the development of specially customized adapters,” said Dr. Janko Schildt, Emperra’s founder and managing director responsible for medicine and technology. “As a result, every insulin-dependent patient can be equipped with Esysta, regardless of whether the insulin comes from Lilly, Sanofi or Novo Nordisk.”
“We are proud that we can use all U-100 insulins in 3 ml cartridges with the development of specially customized adapters,” said Dr. Janko Schildt, Emperra’s founder and managing director responsible for medicine and technology. “As a result, every insulin-dependent patient can be equipped with Esysta, regardless of whether the insulin comes from Lilly, Sanofi or Novo Nordisk.”
“Even the accidental mixing up of basal and bolus Esysta pens and priming (deaeration) can be identified on the Esysta portal. In combination with real insulin application data, I believe as a doctor that this constitutes an evolutionary leap for the digital age of diabetes management,” he said.
Seamless documentation of insulin units and blood glucose levels can be an important element of diabetes treatment, with diabetics needing to measure their blood glucos e several times daily. They need to inject insulin and the associated data needs to be continuously documented and analyzed for optimum metabolic control, which can be a tedious undertaking for both doctor and patient.
e several times daily. They need to inject insulin and the associated data needs to be continuously documented and analyzed for optimum metabolic control, which can be a tedious undertaking for both doctor and patient.
“Often the patients’ entries are difficult to read or simply forgotten. Analyzing the data is laborious and time-consuming,” said Prof. Dr. Klaus Kusterer, a diabetologist from Mannheim, describing many doctors’ everyday experience. “Digital data recording can make a huge contribution to improving diabetes management. Insulin doses and blood glucose levels are automatically recorded using the Esysta system. The data is complete and unadulterated and the patient no longer has to remember to document their data at all.”
HbA1c Reduction Demonstrated Thanks to Digital Diabetes Management
Emperra says that in a study conducted in partnership with a health insurance company, the Esysta system was tested in everyday use by several hundred insulin-dependent diabetics. The study investigators concluded that HbA1c levels — glycated hemoglobin (A1c) which identifies average plasma glucose concentration and provides a longer-term trend of how high blood sugar levels have been over a period of time — could be reduced by nearly 1 percent on average over 12 months.
In patients with significantly higher HbA1c levels at the outset, a reduction of up to 2 percent could be achieved in some cases without elevating the hypoglycemic rate (low blood sugar emergencies), based on the health insurance company’s data. These results are deemed extremely positive, especially with insulin consumption being an important cost factor.
Going forward, Emperra expects that another integrated data transfer solution — the worlds smallest GSM-compatible blood glucose meter — will be ready for release in early 2017, enabling automatic digital transfers to the 868 MHz insulin pen of blood glucose levels and bread units. Emperra says this product is being developed in cooperation with two major international companies for worldwide use in all target groups.
For more information, visit http://www.emperra.com.

